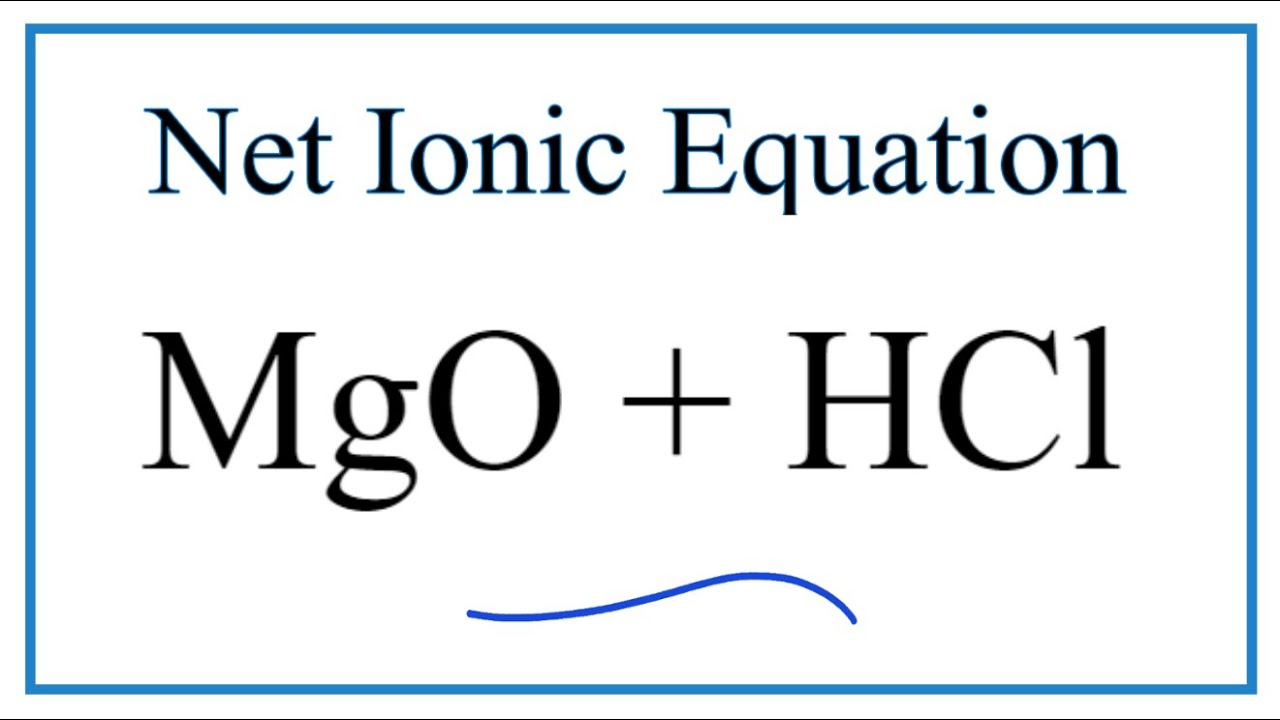Malox, an over-the-counter antacid, contains aluminum hydroxide, Al(OH) 3, and magnesium hydroxides, Mg(OH). What are balanced equations for the reaction of both with stomach acid (HCl)? - Quora

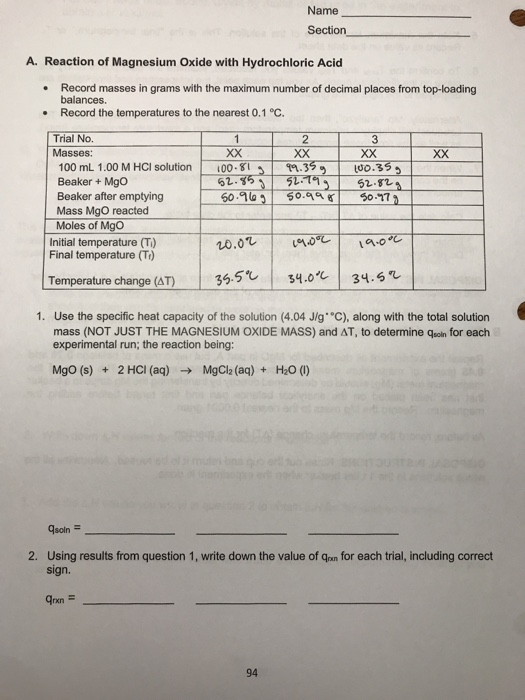
SOLVED: Reaction of Magnesium Oxide with Hydrochloric Acid Trial No Masses: XX XX 100 mL 00 M HCI solution Jno Ghs 91. 5S4 1o.31 Beaker Mgo 52 629 52 ,219 S2,6L) Beaker

What is an oxide? An oxide is a Binary compound of oxygen and another element. M & O Oxides can be classified in two ways – Nature of Oxides Amount of. -

SOLVED: Consider the reaction of magnesium oxide with hydrochloric acid: Mgo (s) 2 HCI (aq) + MgClz (aq) Hz (g) AH -151kI Is the reaction of MgO with HCl exothermic or endothermic?

Write the balanced chemical equations for the following reaction:Calcium hydroxide + Carbon dioxide → Calcium Carbonate + Water.

Ionic equations A chemical equation shows the number of atoms and molecules of the reactants and products. Also shows physical state of reactants and products. - ppt download

SOLVED:Magnesium carbonate, magnesium oxide, and magnesium hydroxide are all white solids that react with acidic solutions. (a) Write a balanced molecular equation and a net ionic equation for the reaction that occurs

Introduction to Acids and Bases. Acid A substance that produces hydrogen ions, H + (aq), when it dissolves in water. Sour-tasting and good conductors. - ppt download