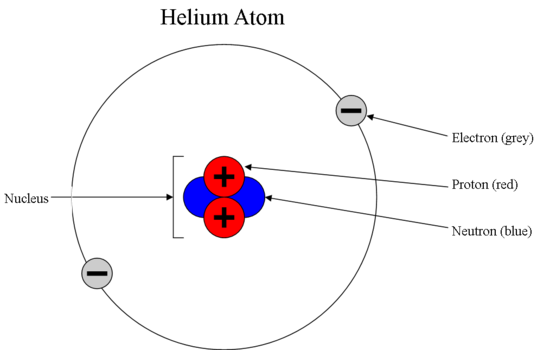
Question Video: Identifying the Energy Level Diagram That Represents the Electronic Configuration of the Magnesium Ion | Nagwa
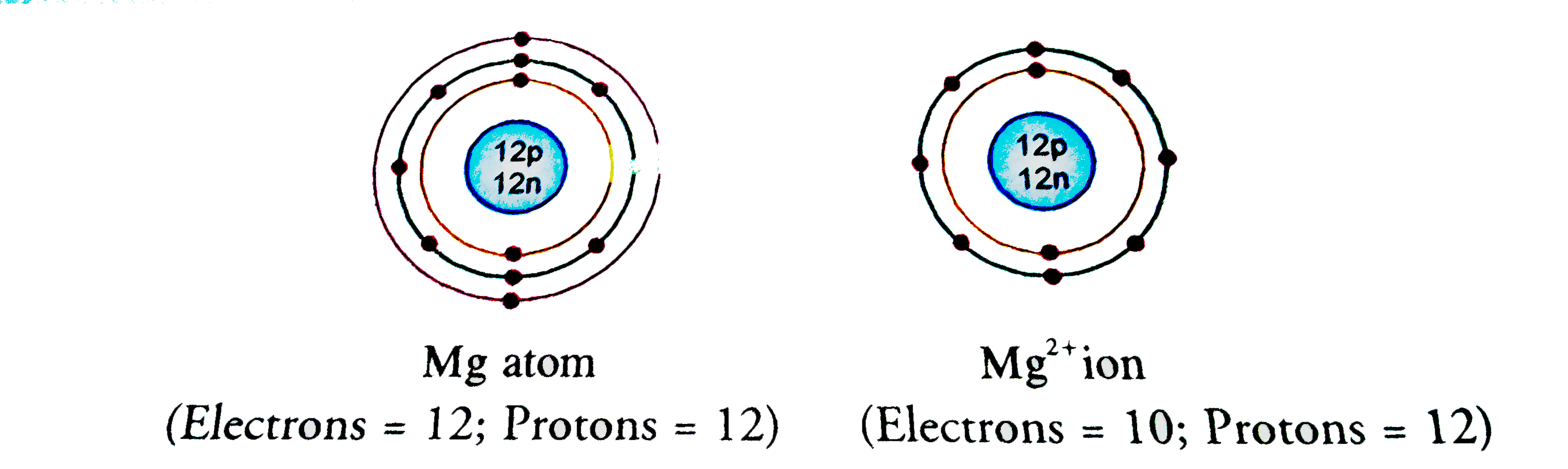
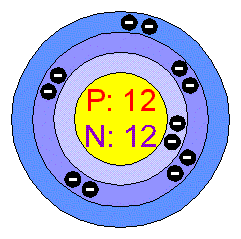
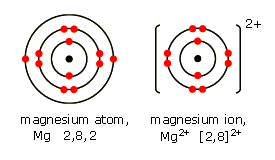
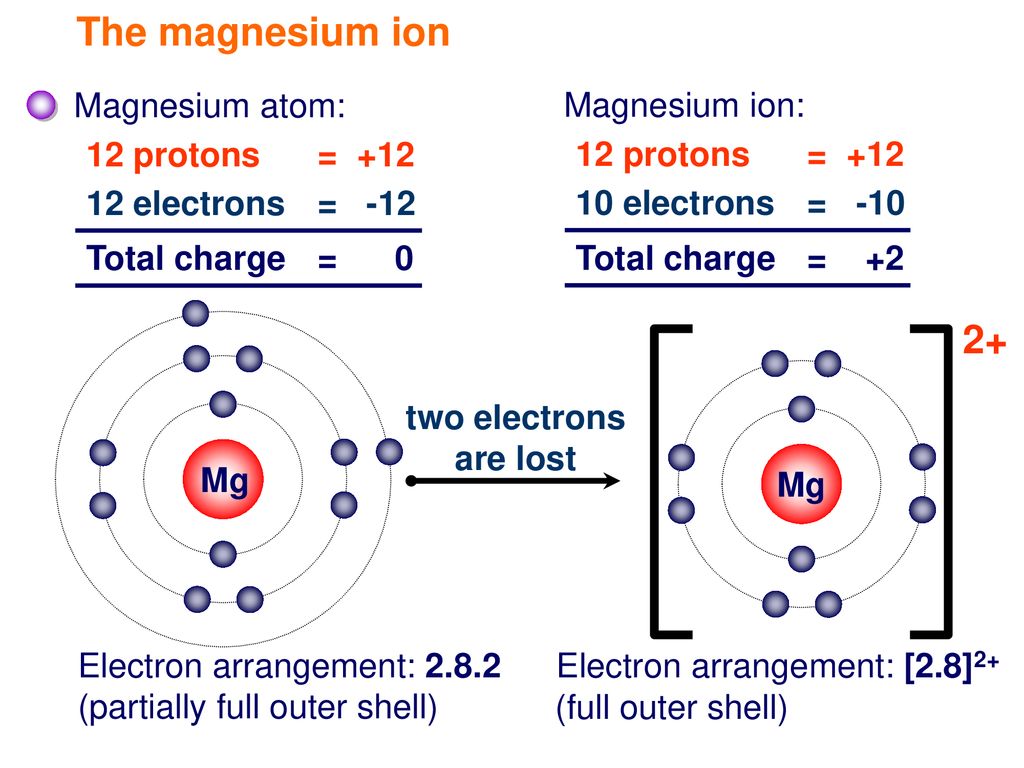
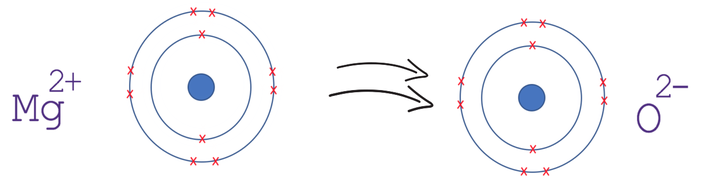
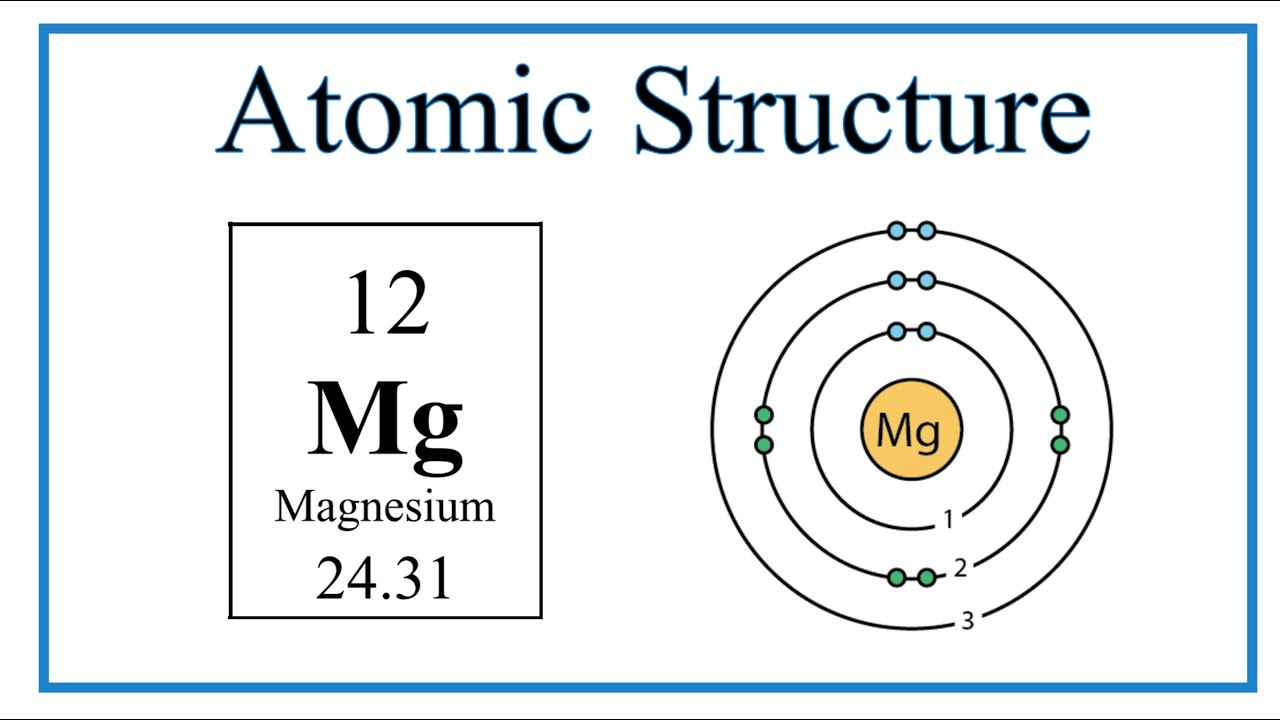
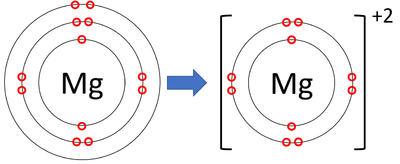
Show the electron distribution in magnesium atom and magnesium ion diagrammatically and also give their atomic numbers.

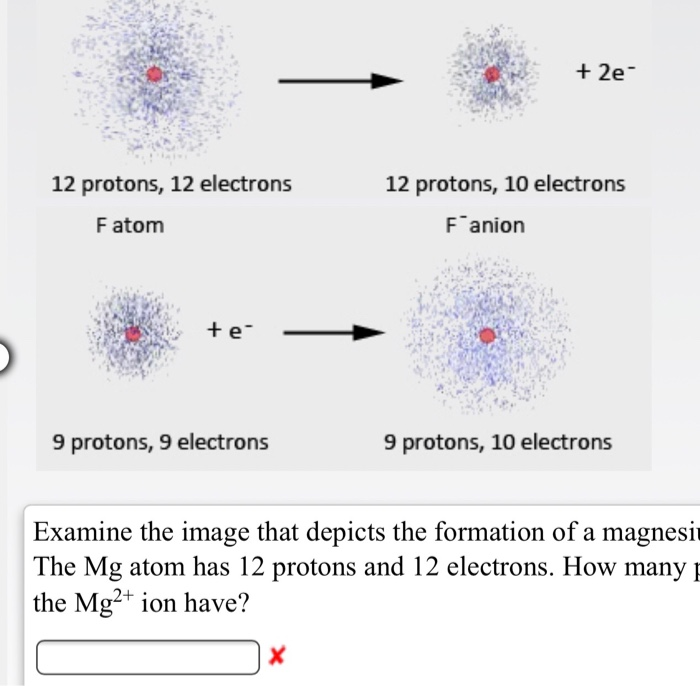
Identify the Mg^(2+) ion from the figure where, n and p represent the number of neutrons and protons respectively.

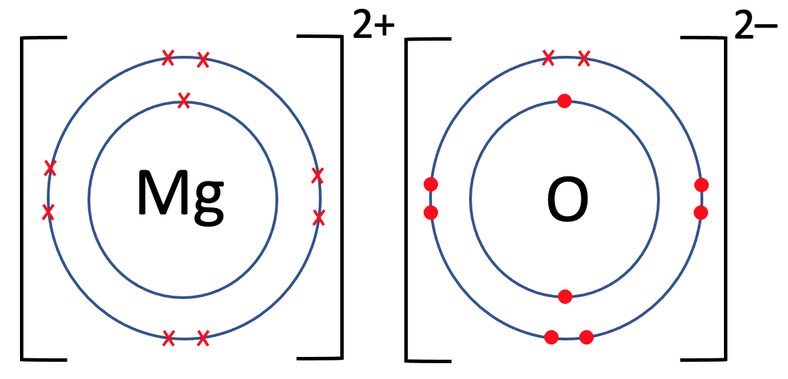
What is the Difference Between Magnesium Atom and Magnesium Ion | Compare the Difference Between Similar Terms

Identify the Mg^2 + ion from the Fig.4.1 where \"n\" and \"p\" represent the number of neutrons and the number of protons respectively.

Identify the Mg2+ ion from Fig. where n and p represent the number of neutrons and protons respectively.

What is the Difference Between Magnesium Atom and Magnesium Ion | Compare the Difference Between Similar Terms

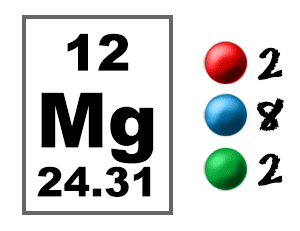
SOLVED: Complete the following table Name of element or ion #of neutrons #of #of protons electrons 14 12 12 140 8 Atomic Mass Symbol Magnesium Radiun 06 Mg ee Ra 226 3