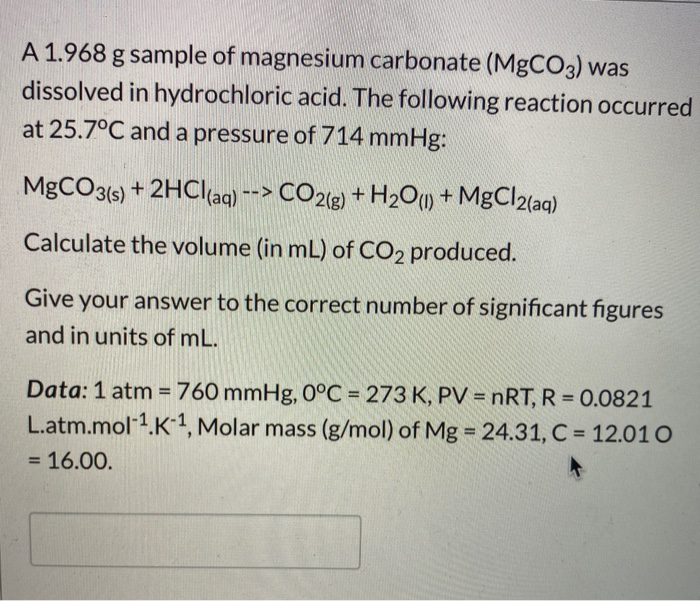
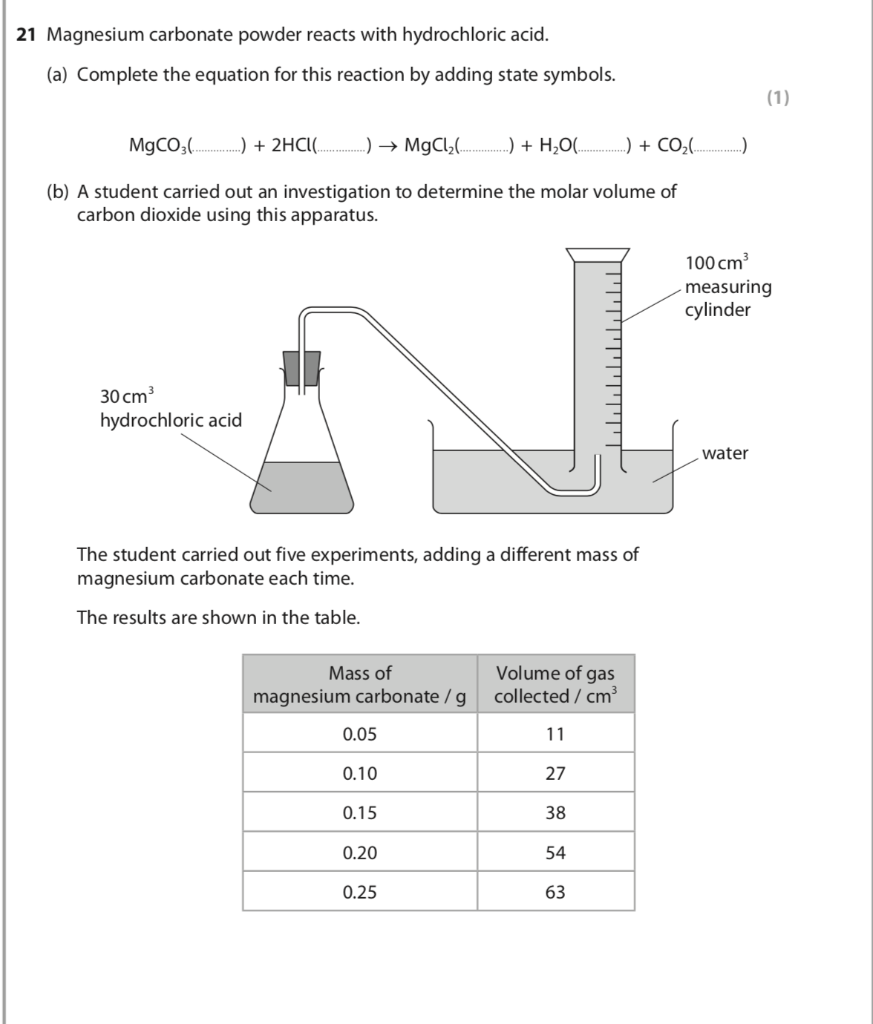
SOLVED: Geologists identify carbonate minerals by reaction with acids Dolomite , for example , contains magnesium carbonate , which reacts with hydrochloric acid by Ihe following reaction: MgCOs (s) + HCl(aq) ,MgClz (

SOLVED:The mineral dolomite contains magnesium carbonate. This reacts with hydrochloric acid. MgCO3(s)+2 HCl(aq) →CO2(g)+MgCl2(aq)+H2 O(ℓ) (a) Write the net ionic equation for this reaction and identify the spectator ions. (b) What type

filosoffen.dk - what is metformin 500 mg used for | Commit error. what is the word equation for calcium carbonate and hydrochloric acid congratulate

Magnesium carbonate reacts with hydrochloric acid to form magnesium chloride, carbon dioxide and water Translate and balance the equation - Science - Chemical Reactions and Equations - 12554199 | Meritnation.com

balanced equation for the following a)megnesium carbonate reacts with HCL acid to produce megnesium - Brainly.in
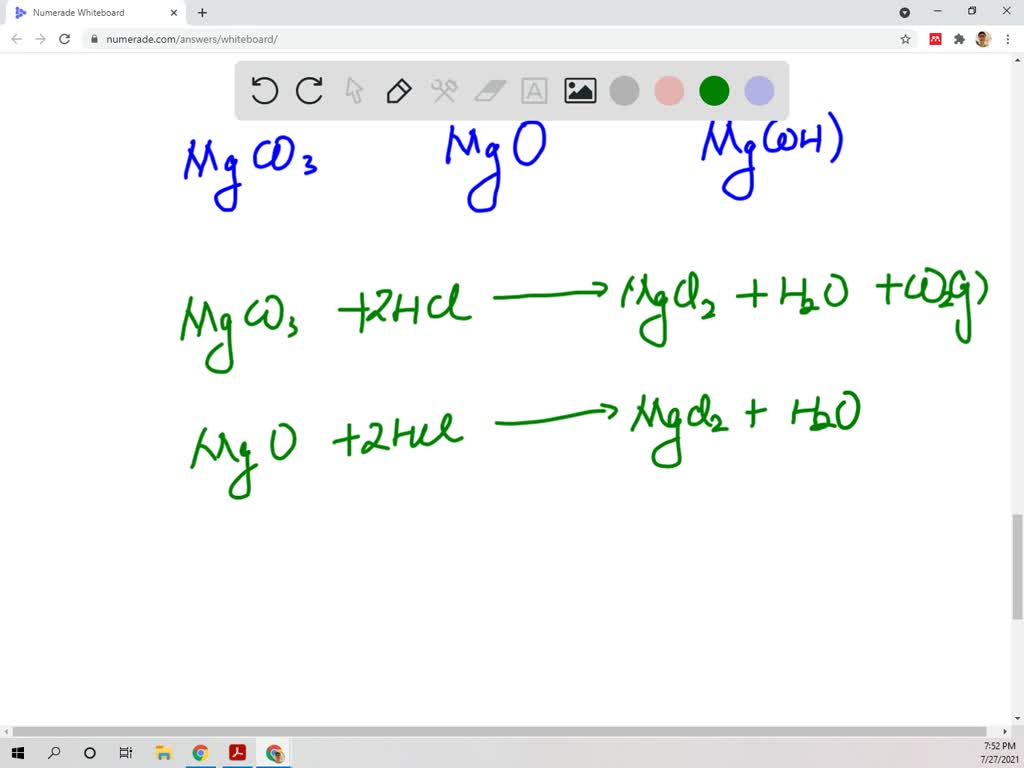
SOLVED: Magnesium carbonate, magnesium oxide, and magnesium hydroxide are all white solids that react with acidic solutions. (a) Write a balanced molecular equation and a netionic equation for the reaction that occurs

For Your Research. The Four Research Questions 1.What is the chemistry (including an equation) of the process? 2.What are the factors that impact on the. - ppt download