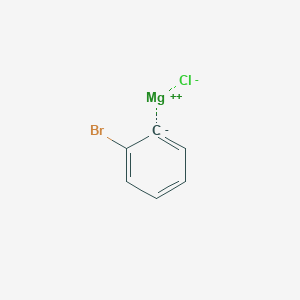![4-(1-Piperidinylmethyl)phenyl]magnesium bromide, 0.25M solution in THF, AcroSeal™, Thermo Scientific 4-(1-Piperidinylmethyl)phenyl]magnesium bromide, 0.25M solution in THF, AcroSeal™, Thermo Scientific](https://www.thermofisher.com/TFS-Assets/CCG/Chemical-Structures/chemical-structure-cas-480424-83-7.jpg-650.jpg)
4-(1-Piperidinylmethyl)phenyl]magnesium bromide, 0.25M solution in THF, AcroSeal™, Thermo Scientific
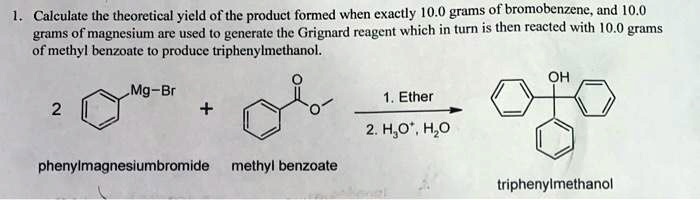
SOLVED: Calculate the theoretical yield of the product formed when exactly 10.0 grams of bromobenzene and 0.0 grams of magnesium are used to generate the Grignard reagent, which in turn is then

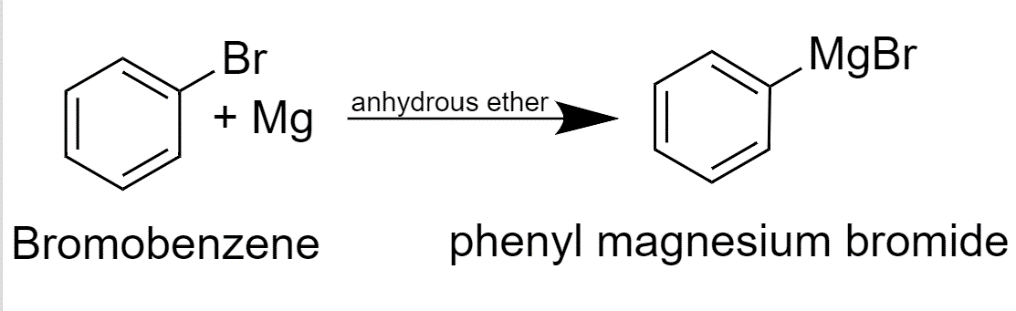
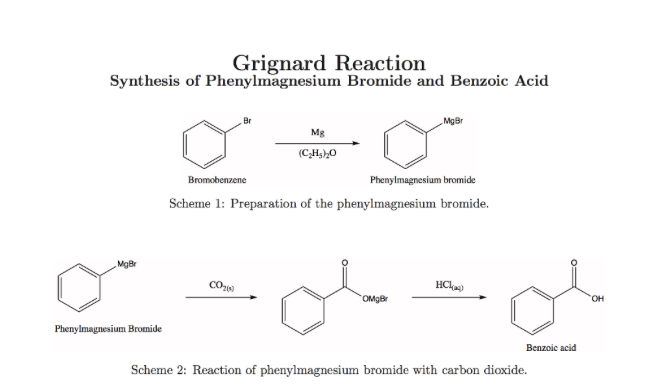
A Grignard reagent was prepared by reacting bromobenzene with magnesium in diethyl ether. Carbon dioxide gas was then bubbled through the solution, resulting in the formation of a solid organic produc
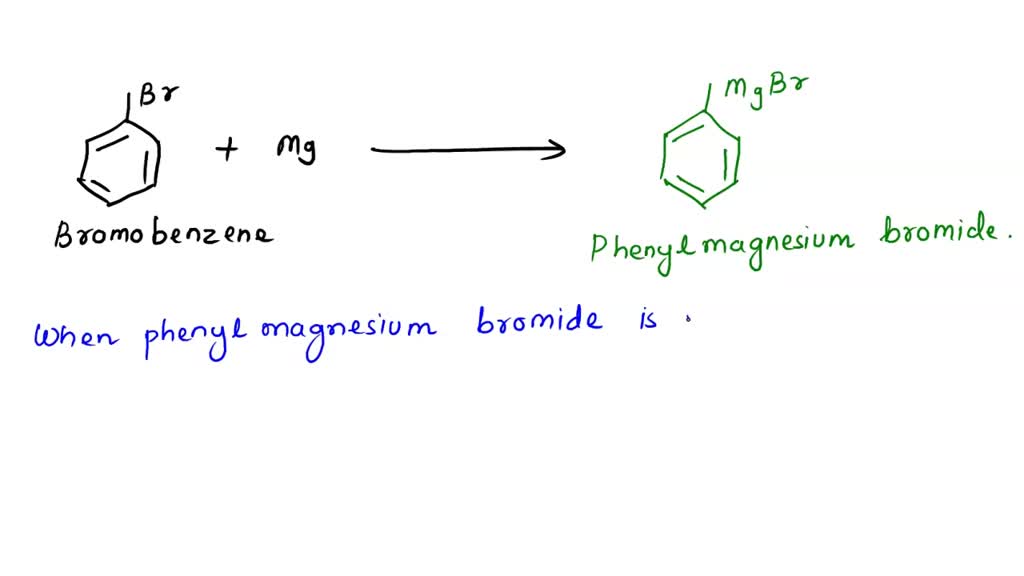
SOLVED: A student accidently used methanol as the reaction solvent for this experiment. Once the bromobenzene and the magnesium were mixed, a vigorous reaction took place and a volatile liquid was isolated

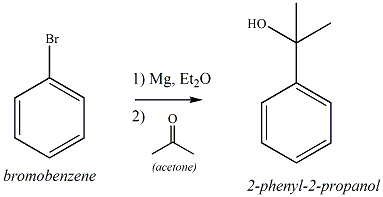
How could you convert bromobenzene into benzoic acid, C_6H_5CO_2H? (More than one step is required.) | Homework.Study.com

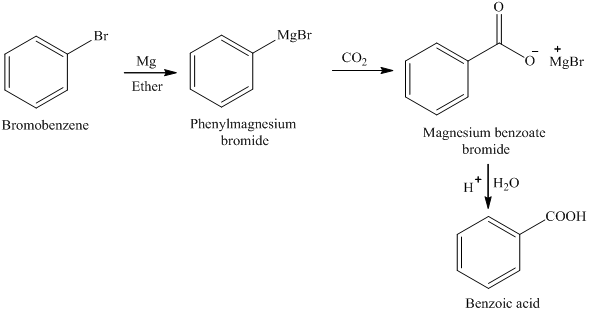


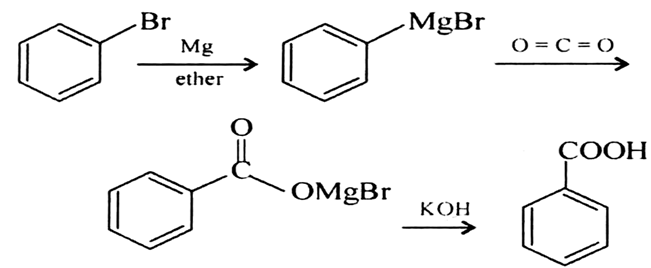
![ch216sp12syllabus [licensed for non-commercial use only] / Experiment 7 ch216sp12syllabus [licensed for non-commercial use only] / Experiment 7](http://ch216sp12syllabus.pbworks.com/f/1338819727/GirgnardRxtnScheme.png)
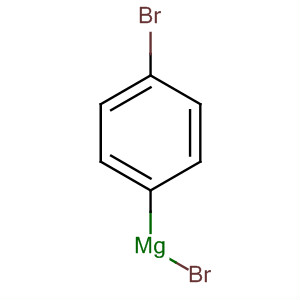

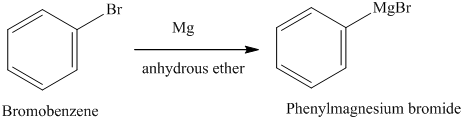
![ch216sp12syllabus [licensed for non-commercial use only] / Experiment 7 ch216sp12syllabus [licensed for non-commercial use only] / Experiment 7](http://ch216sp12syllabus.pbworks.com/f/1338819721/Grignardreagent.png)